for clear pic pls click below link.
Every time there’s a surgery, however colorful attire the surgeons may have donned, the moment he or she enters an operation theatre, he wears that same green or blue overalls and the same cap. Wonder why he does that? Well, there are scientific reasons for it.
History Speaks
First, let’s go back to the year 1914.

White overalls of the surgeons during the 1900s
Before the blue or green dress code the surgeons used to wear white clothes during a surgery. However, according to an article published in 1998’s Today’s Surgical Nurse, in 1914 this trend came to an end with the help of a reputed doctor who introduced the green and blue overalls.
Blinding White Overalls
One of the scientific reasons behind the use of green or blue overalls is that surgeons may get blinded by the completely spotless white overalls once they shift their gaze from the dark red color of the blood to the white clothing of his or her colleagues.
One also experiences the same thing when he or she goes outside in winter and sees the sun’s reflection on the snow.
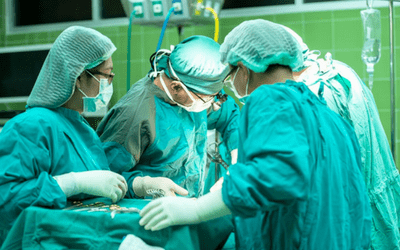
Surgeons now wear green or blue overalls
Seeing Illusions During a Surgery is a Big 'NO'
The surgeons may also experience certain green illusions after gazing at blood for so long a time. Now, this tends to intensify if his or her co-surgeons wear white overalls as he or she may see distracting green illusions on the white surfaces (quite similar to the after effects of a camera flash). Whereas if it’s a color that is a complete opposite to red then, the illusions are greatly reduced.
Because Green and Blue are the Opposites of Red
While performing a surgery the surgeon mostly sees the color of the blood, tissues and muscles that are red and pink. After a point he is not able to sense the difference between the two as both the colors are associated. But looking at contrasting colors like blue or green helps the doctor to differentiate colors and concentrate on the patient and his or her anatomy.
Visibly Accurate
The green and blue overall, therefore, provides the surgeons with a visual accuracy enhancing their sensitivity to the different shades of red present in the patient’s blood. This not only helps the surgeons to prevent mistakes during surgery but also to pay more attention to their patient’s anatomy.
thankx. https://www.vaidam.com/








